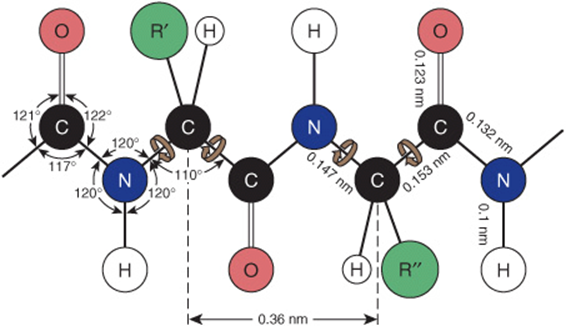
(1966) except that the origin is now at the center, instead of at the lower left hand corner. Note: this diagram is identical with that of Edsall et al. Right handed α helix (α-poly( L-alanine))Īntiparallel-chain pleated sheet (β-poly( L-alanine))Ī For a fully extended chain φ = ψ = &oega = +180°.įigure 6: Typical conformatial map ( Ramachandran et al., 1963) transposed into the standard conventions. Table II: Approximate Torsion Angles for Some Regular Structures. a,bĪ trans to N i-H i is the same as cis to N i-C i-1 trans to C i-O i is the same as cis to C i-N i+1, (see Figure 5).ī For the description of D-amino acids, interchange C α-H and C α-R in the table. Table I Main-Chain Torsion Angles for Various Conformations in Peptides of L-Amino Acids. (v) Figure 6 is a typical conformational map ( φ-ψ plot) using the rules enunciated above. It is noteworthy that a right-handed α helix has negative torsion angles. (iv) Table II gives values of φ and ψ for various well-known regular structures. The case of φ = ψ = 0° would involve the relations indicated in Table I. (iii) A "fully extended" polypeptide chain is characterized by φ = ψ = ω = +180°. ω ~ 180° is the value which is generally found (i.e., the trans conformation), (ii) Owing to the partial double-bond character of, it is normally possible for ω to assume values only in the A neighborhood of 0 or 180°. (This statement is precisely correct only if the peptide bond is exactly planar, which is not generally the case in experimentally determined structures). The new designation of angles may be derived from the old by adding 180° to, or subtracting 180° from, the latter. (i) This convention differs from that proposed by Edsall et al. The chain is shown in a fully extended conformation (φ i = ψ i = ω i = 180°), and the residue illustrated is L. The limits of a residue are indicated by dashed lines, and recommended notations for atoms and torsion angles are indicated. In accordance with Rules 1.6 and 2.1.1, these torsion angles are ascribed zero values for eclipsed conformations of the main-chain atoms N, C α, and C, that is, for the so-called cis conformations (see Table I).įigure 5: Perspective drawing of a section of polypeptide chain representing two peptide units. The symbols φ i, ψ i, and ω i are used to denote torsion angles of bonds within the i th residue in the case of φ and ψ and between the i th and ( i +1)th residues in the case of ω specifically, φ i refers to the torsion angle of the sequence of atoms C i-1, N i, C i α, C i ψ ito the sequence N i, C i α, C i, N i+l and ω i to the sequence C i α, C i, N i+1, C i+1 α (see Figure 5). The principal torsion angle describing rotation about N-C α is denoted by φ, that describing rotation about C α-C is denoted by ψ, and that describing rotation about C-N is denoted by ω. Bond lengths are written b(C i,N i+l), etc.ģ.2.1. 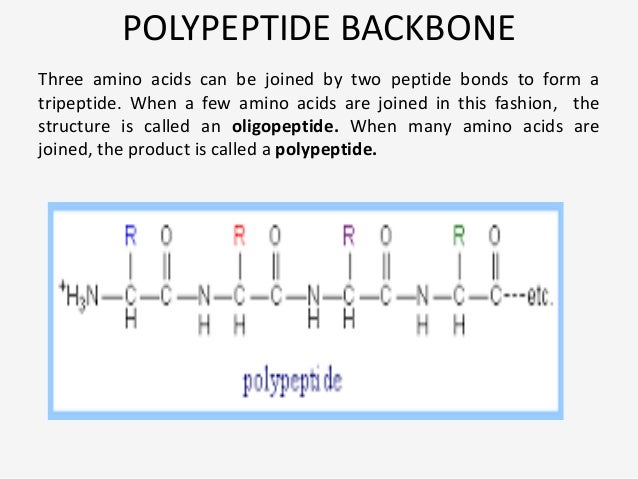
The Main Chain (or Polypeptide Backbone)īonds between main-chain atoms are denoted by the symbols of the two atoms terminating them, e.g., N i-C i α, C i α-C i, C i-N i+1, C i-O i, N i-H i. The Sequence Rules and Choice of Torsion Angle Polypeptide Conformations 3 IUPAC-IUB Commission on Biochemical Nomenclature (CBN)Ībbreviations and Symbols for the Description of the Conformation of Polypeptide ChainsĬontinued from 2.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
